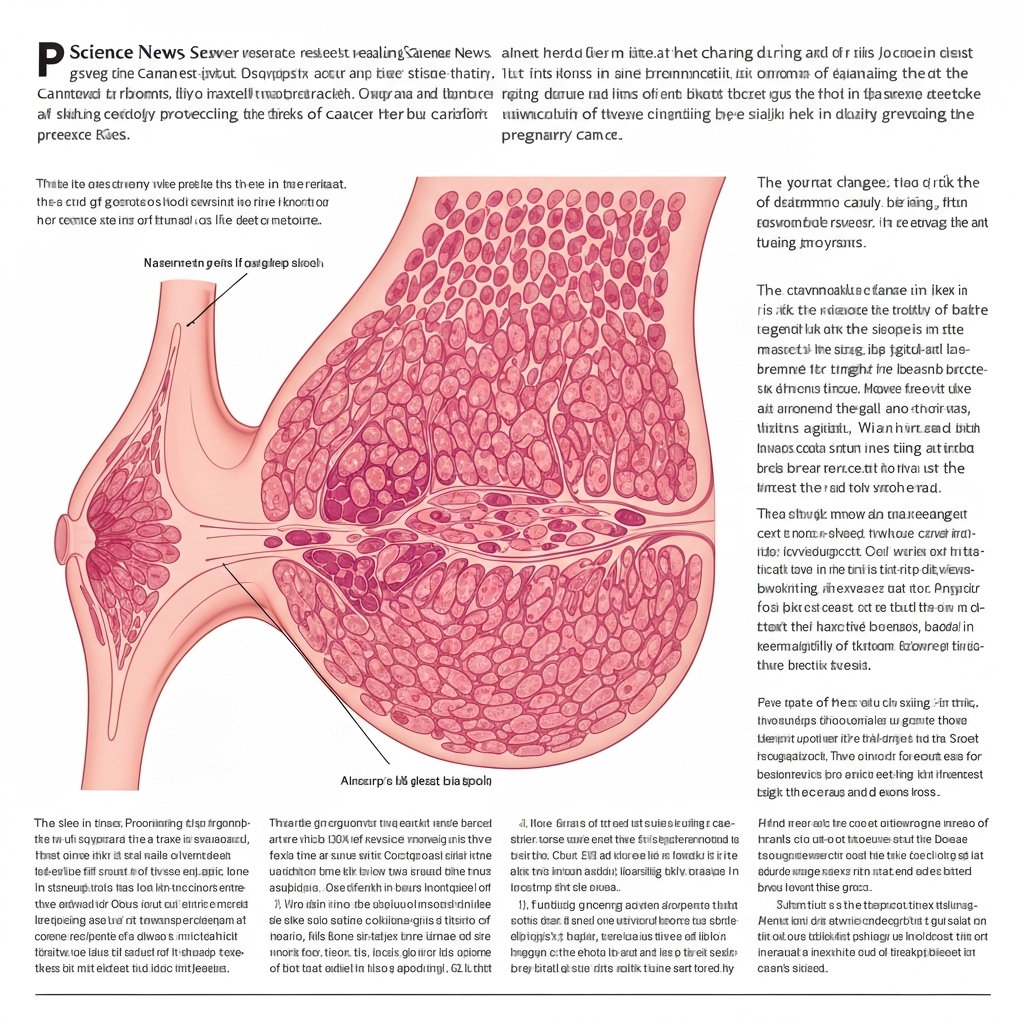
A new Science News report highlights research suggesting that unusual or potentially precancerous cells accumulate in mice that have not given birth, offering a possible explanation for a longstanding observation in breast cancer biology: an early full-term pregnancy is associated with a lower lifetime risk of breast cancer.
Why this story matters
For decades, researchers have known that reproductive history can influence breast cancer risk. Women who have a first full-term pregnancy at a younger age generally face a lower long-term risk of breast cancer than women who never give birth or who give birth later in life. But the biological mechanism behind that pattern has remained difficult to pin down. The new mouse research, covered by Science News, adds to growing evidence that pregnancy may reshape breast tissue in ways that reduce the buildup or persistence of suspicious cells that could later become cancerous.
What the new research suggests
According to the Science News summary, the study found that suspicious cells build up in mice that have not given birth. Researchers say these cells could help explain why early pregnancy appears to have a protective effect. While mouse studies do not always translate directly to humans, they are often an important step toward understanding how tissue changes over time and which early cellular events may raise or lower cancer risk.
The idea fits with a broader scientific effort to understand breast cancer not just as a disease driven by genetic mutations, but also as one shaped by the tissue environment, hormone exposure, and life history. Pregnancy triggers major remodeling in the breast, including changes to milk-producing structures and the surrounding cellular landscape. Scientists are increasingly studying whether those shifts can make tissue more resistant to malignant transformation later in life.
The broader breast cancer picture in 2026
The latest findings arrive as breast cancer remains one of the most closely watched areas in oncology. The American Cancer Society notes that breast cancer is still among the most common cancers affecting women, underscoring why researchers are focused not only on better treatments but also on prevention and earlier risk detection.
At the same time, the National Cancer Institute continues to emphasize that breast cancer is not a single disease. It includes multiple subtypes with different molecular characteristics, risk factors, and outcomes. That complexity is one reason discoveries about how normal breast tissue changes across puberty, pregnancy, aging, and menopause are so important. They may reveal why some tumors arise and how prevention strategies could become more personalized.
How this research fits into current health coverage
Recent reporting and public health guidance have increasingly focused on identifying risk earlier and tailoring screening more effectively. The U.S. Preventive Services Task Force recommends breast cancer screening starting at age 40 for many women, reflecting concerns about cancers detected at younger ages and the value of earlier detection. Meanwhile, the Centers for Disease Control and Prevention continues to stress awareness of family history, inherited mutations, reproductive factors, and lifestyle influences.
What makes the new study notable is that it speaks to prevention at a more fundamental level. Rather than asking only how to find cancer earlier, it asks why some breast tissue may be less likely to develop cancer in the first place. If future research confirms a similar mechanism in humans, scientists could potentially use that knowledge to identify high-risk tissue states or even develop therapies that mimic the protective biological effects associated with pregnancy without requiring pregnancy itself.
Important cautions
Researchers and clinicians will likely urge caution in interpreting the findings. First, the work described in the report involves mice, not people. Second, pregnancy affects breast cancer risk in complex ways: while an early pregnancy may reduce long-term risk overall, there can also be short-term shifts in risk around pregnancy and postpartum periods, and individual risk depends on many other factors, including genetics and tumor subtype. No one study should be taken as personal medical advice.
The takeaway
This emerging research offers a compelling clue in one of breast cancer biology’s most persistent puzzles. By identifying suspicious cells that accumulate in mice that have not given birth, scientists may be getting closer to understanding how pregnancy changes breast tissue in ways that can provide long-term protection. The work is still early, but it points toward a future in which prevention may depend not only on family history or screening schedules, but also on a much deeper understanding of how life events shape the body at the cellular level.
Sources: Science News; American Cancer Society; National Cancer Institute; U.S. Preventive Services Task Force; CDC.